  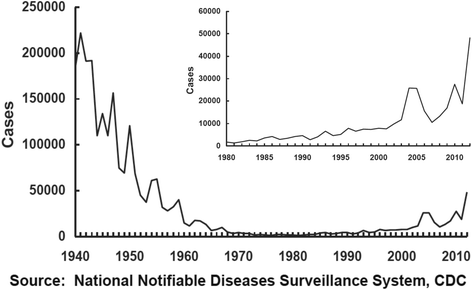
However, few published studies have evaluated the safety of Tdap vaccine in pregnant women. 4 - 6 Most recently, in 2012, the Centers for Disease Control and Prevention (CDC) Advisory Committee on Immunization Practices (ACIP) recommended tetanus, diphtheria, and acellular pertussis (Tdap) vaccination for all pregnant women during each pregnancy regardless of prior immunization status. Many public health strategies have been recommended to decrease the burden of pertussis in neonates and infants. 1 - 3 Neonates and infants are at increased risk of pertussis-related hospitalization and death compared with older children and adults. Pertussis (whooping cough) is a vaccine-preventable illness that has been increasing in incidence over the past decade in the United States. These findings suggest that relatively recent receipt of a prior tetanus-containing vaccination does not increase risk after Tdap vaccination in pregnancy. 
Small for gestational age delivery occurred in 9.0% of women less than 2 years before (adjusted RR, 0.99 P = .88) and 8.7% of women 2 to 5 years before (adjusted RR, 0.96 P = .45) a prior tetanus-containing vaccine compared with 9.1% of controls.Ĭonclusions and Relevance Among women who received Tdap vaccination during pregnancy, there was no increased risk of acute adverse events or adverse birth outcomes for those who had been previously vaccinated less than 2 years before or 2 to 5 years before compared with those who had been vaccinated more than 5 years before. Preterm delivery occurred in 6.6% of women receiving Tdap in pregnancy less than 2 years before (adjusted RR, 1.15 P = .08) and 6.4% two to 5 years before (adjusted RR, 1.06 P = .33) a prior tetanus-containing vaccine compared with 6.8% of controls. For example, local reactions occurred at a rate (per 10 000 women) of 4.2 in those who received Tdap in pregnancy less than 2 years before (adjusted risk ratio, 0.49 P = .35) and 7.0 two to 5 years before (adjusted RR, 0.77 P = .59) a prior tetanus-containing vaccine compared with 11.2 in controls. Results There were no statistically significant differences in rates of medically attended acute adverse events or adverse birth outcomes related to timing since prior tetanus-containing vaccination. Women who were vaccinated with Tdap in pregnancy and had a prior tetanus-containing vaccine more than 5 years before served as controls. Main Outcomes and Measures Acute adverse events (fever, allergy, and local reactions) and adverse birth outcomes (small for gestational age, preterm delivery, and low birth weight) were evaluated. Objective To determine whether receipt of Tdap vaccine during pregnancy administered in close intervals from prior tetanus-containing vaccinations is associated with acute adverse events in mothers and adverse birth outcomes in neonates.ĭesign, Setting, and Participants A retrospective cohort study in 29 155 pregnant women aged 14 through 49 years from January 1, 2007, through November 15, 2013, using data from 7 Vaccine Safety Datalink sites in California, Colorado, Minnesota, Oregon, Washington, and Wisconsin.Įxposures Women who received Tdap in pregnancy following a prior tetanus-containing vaccine less than 2 years before, 2 to 5 years before, and more than 5 years before. However, safety data on repeated Tdap vaccination in pregnancy is lacking. Importance The Advisory Committee on Immunization Practices (ACIP) recommends the tetanus, diphtheria, and acellular pertussis (Tdap) vaccine for pregnant women during each pregnancy, regardless of prior immunization status. Shared Decision Making and Communication.Scientific Discovery and the Future of Medicine.Health Care Economics, Insurance, Payment.Clinical Implications of Basic Neuroscience.Challenges in Clinical Electrocardiography. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
